News & Announcements
MS Thesis Defense - Max Campbell
MS Thesis Defense - Max Campbell
Advisor: Peter Chien
Monday April 8
4pm in LSL N210
Jessica McGory Dissertation Defense
Jessica McGory Dissertation Defense
Title: "The role of intrinsically disordered proteins in the regulation of chromosome segregation"
Advisor: Thomas Maresca
Thursday April 11, 2024
1:00 PM
LGRT 1681
Zoom link: https://umass-amherst.zoom.us/j/97898155068
Successful Biotech Training Program Retreat in Boston
Successful Biotech Training Program Retreat in Boston
The Biotech Training Program (BTP) had an excellent time at our 3-day retreat in the Biotech Hub in Boston. We visited Ginko Bioworks, Dewpoint Therapeutics and experienced an excellent tour of Vertex Cell Therapies group with MCB alumni Brad Pearse!
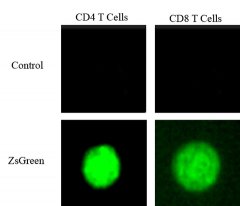
Researchers at UMASS Amherst Shed Light on How Tumor Cells Outwit the Body's Immune System
Researchers at UMASS Amherst Shed Light on How Tumor Cells Outwit the Body's Immune System
In a first-of-its-kind research breakthrough, a team of scientists at the University of Massachusetts Amherst has analyzed and described what they call the “mosquito effect,” which sheds light on how certain pathogens, such as cancerous tumor cells, can outwit the body’s immune system.
Just as mosquitoes ingest their host’s blood, the immune system’s T cells incorporate cytoplasmic material from tumors into their own cytoplasm. While it has long been known that many kinds of cells can transfer cellular material from one to another, the transfer of the cytoplasm has never been observed in T cells. Subsequent single-cell RNA (scRNA) sequencing shows that cytoplasm from tumor cells alters the machinery responsible for protein coding in the host T cell. The research, reported recently in the journal Frontiers in Immunology, is a major step forward in understanding how tumors can successfully evade the immune system, and thus a step toward more effective treatments.
To make the discovery, Pobezinsky and his team, including first author Kaito Hioki, graduate student in the MCB program at UMass Amherst, and Elena Pobezinskaya, research assistant professor also in veterinary and animal sciences at UMass and co-senior author of the paper, engineered tumor cells to produce an ultrabright fluorescent protein called ZsGreen. They then introduced the green-glowing tumor cells into a mouse model. After eight days, the model’s tumor-infiltrating immune cells were gathered and analyzed using state-of-the-art equipment in the Flow Cytometry lab at UMass Amherst’s Institute for Applied Life Sciences. Read more
UMASS Amherst Researchers Crack the Cellular Code on Protein Folding, Opening New Therapeutic Avenues for Many Diseases
UMASS Amherst Researchers Crack the Cellular Code on Protein Folding, Opening New Therapeutic Avenues for Many Diseases
While we often think of diseases as caused by foreign bodies—bacteria or viruses—there are hundreds of diseases affecting humans that result from errors in cellular production of its proteins. A team of researchers led by the University of Massachusetts Amherst recently leveraged the power of cutting-edge technology, including an innovative technique called glycoproteomics, to unlock the carbohydrate-based code that governs how certain classes of proteins form themselves into the complex shapes necessary to keep us healthy.
The research, published in the journal Molecular Cell, explores members of a family of proteins called serpins, which are implicated in a number of diseases. The research is the first to investigate how the location and composition of carbohydrates attached to the serpins ensure that they fold correctly. Serious diseases—ranging from emphysema and cystic fibrosis to Alzheimer’s disease—can result when the cellular oversight of protein folding goes awry. Identifying the glyco-code responsible for high-fidelity folding and quality control could be a promising way for drug therapies to target many diseases.
The discovery of the carbohydrate-based chaperone system in the ER was due to the pioneering work that Daniel Hebert, professor of biochemistry and molecular biology at UMass Amherst and one of the paper’s senior authors, initiated as a postdoctoral fellow in the 1990s. “The tools we have now, including glycoproteomics and mass spectrometry at UMass Amherst’s Institute for Applied Life Sciences, are allowing us to answer questions that have remained open for over 25 years,” says Hebert. “The lead author of this new paper, Kevin Guay, is doing things I could only dream of when I first started.” Read more
Jun-Goo Kwak PhD Dissertation Defense
Jun-Goo Kwak PhD Dissertation Defense
Title: "“IN VITRO BONE MARROW MODELS TO INTERROGATE THE IMPACTS OF
SEQUESTRATION AND SECRETION BY BONE MARROW ADIPOCYTES”
Advisor: Jungwoo Lee
LSL N410
January 11th at 1:00 pm
Navya Sebastian Masters Thesis Defense
Navya Sebastian Masters Thesis Defense
Title:"Understanding the Functional Impact of Disease-Associated Phosphorylation Sites on the Neurodegenerative Protein Tau"
Advisor: Jennifer Rauch
December 19th at 12:30pm in LSL N410
Parag Juvale PhD Thesis Defense
Parag Juvale PhD Thesis Defense
Title: "Postsynaptic Changes in Inhibitory Signaling are not Necessary for Experience-Dependent Maintenance of the Visual System"
Advisor: Sarah Pallas
December 20, at 12 pm
Location: Room 222, Morrill
Olivia Macrorie MS Thesis defense
Olivia Macrorie MS Thesis defense
Title: Investigating the role of Got2 in murine organogenesis and placental development
Advisor: Kimberly D Tremblay
Monday December 18 at 1pm in ISB 221